This web page was produced as an assignment for Genetics 677, an undergraduate course at UW-Madison.
String was used to produce all of the following protein interaction networks.
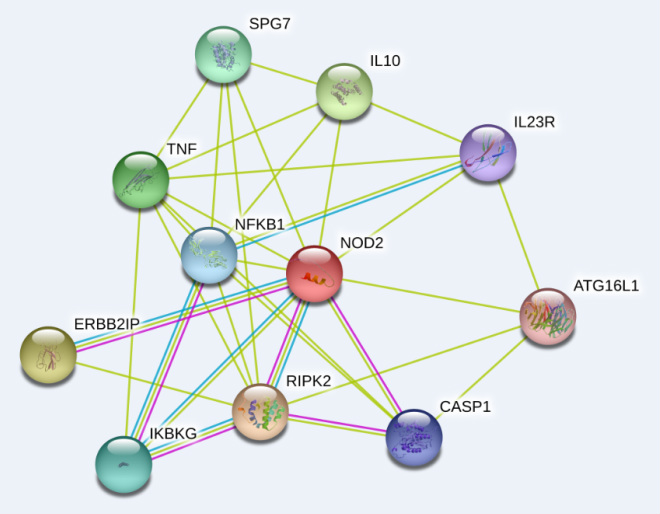
Human NOD2 interaction network
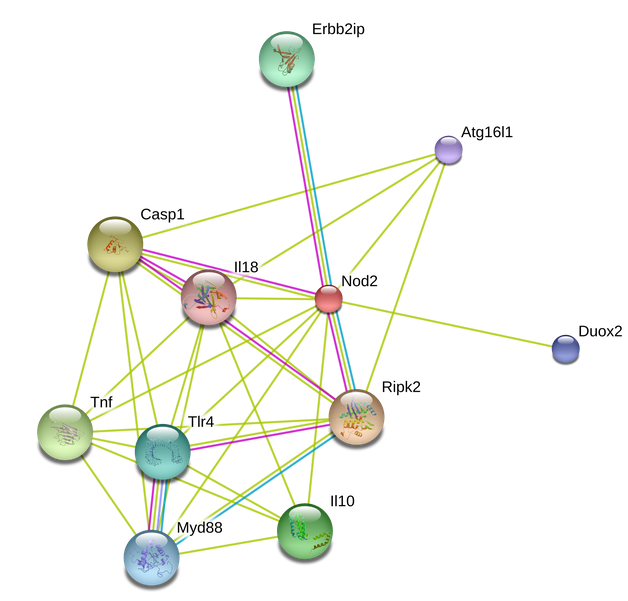
Mouse Nod2 Interaction
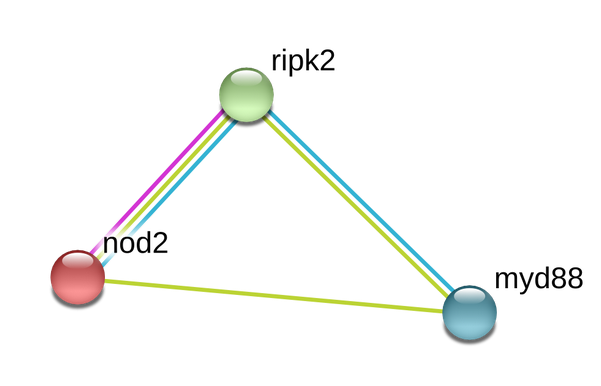
Zebrafish Interactions
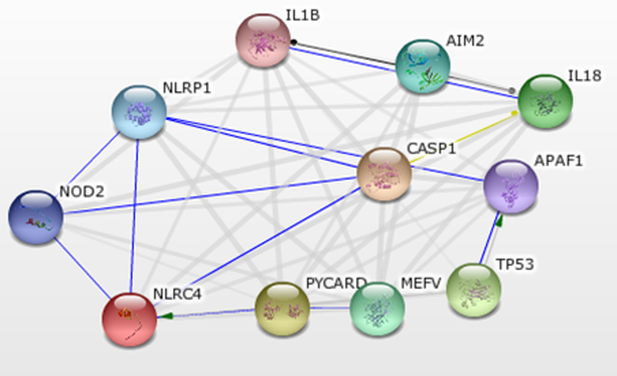
Human NOD2 Binding Partners
Analysis
Comparing human, mouse, and zebrafish NOD2 interactomes confirms that mouse NOD2 is more closely related to Human NOD2, suggesting that mouse models of Crohn's Disease would be more accurate. Unfortunately, the NLRP-NOD2 and NLRC4-NOD2 interactions have not yet been shown in mice, but given homology it is likely that these interactions are also taking place.
Figure 4 highlights interactions of NOD2 in which actual physical contact is involved. Note that Figure 1 incorporates connections based solely on text mining, while Figure 1 only includes compounds that have shown physical interaction experimentally. We focus our attention particularily on NLRP1 and NLRC4(CLAN). Literature review showed that both NLRP1 and NLRC4 interacted with NOD2 to form hetero-oligomers [1,2]. Such oligomers behave differently than NOD2 hetero-oligomers and may be involved in differential bacteria response. Disruption of NOD2, as is the case in Crohn's Disease, would disrupt such hetero-oligomers as well. Analysis of the expression and activity of these directly associating proteins in CD vs control patients may provide insights into CD and thus is a good avenue for future study.
Figure 4 highlights interactions of NOD2 in which actual physical contact is involved. Note that Figure 1 incorporates connections based solely on text mining, while Figure 1 only includes compounds that have shown physical interaction experimentally. We focus our attention particularily on NLRP1 and NLRC4(CLAN). Literature review showed that both NLRP1 and NLRC4 interacted with NOD2 to form hetero-oligomers [1,2]. Such oligomers behave differently than NOD2 hetero-oligomers and may be involved in differential bacteria response. Disruption of NOD2, as is the case in Crohn's Disease, would disrupt such hetero-oligomers as well. Analysis of the expression and activity of these directly associating proteins in CD vs control patients may provide insights into CD and thus is a good avenue for future study.
References
[1] Damiano JS, Oliveira V, Welsh K, Reed JC. Heterotypic interactions among NACHT domains: implications for regulation of innate immune responses. Biochem J. 2004;381(Pt 1):213-9.
[2] Hsu, L.-C., S. R. Ali, S. McGillivray, P.-H. Tseng, S. Mariathasan, E. W. Humke, L. Eckmann, J. J. Powell, V. Nizet, V. M. Dixit, and M. Karin. "A NOD2-NALP1 Complex Mediates Caspase-1-dependent IL-1 Secretion in Response to Bacillus Anthracis Infection and Muramyl Dipeptide." Proceedings of the National Academy of Sciences 105.22 (2008): 7803-808. Print.
[3] String
[2] Hsu, L.-C., S. R. Ali, S. McGillivray, P.-H. Tseng, S. Mariathasan, E. W. Humke, L. Eckmann, J. J. Powell, V. Nizet, V. M. Dixit, and M. Karin. "A NOD2-NALP1 Complex Mediates Caspase-1-dependent IL-1 Secretion in Response to Bacillus Anthracis Infection and Muramyl Dipeptide." Proceedings of the National Academy of Sciences 105.22 (2008): 7803-808. Print.
[3] String