This web page was produced as an assignment for Genetics 677, an undergraduate course at UW-Madison.
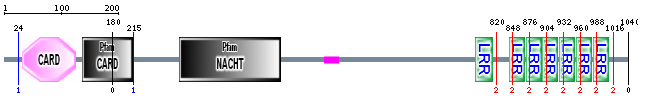
NOD2 Domains and Structure
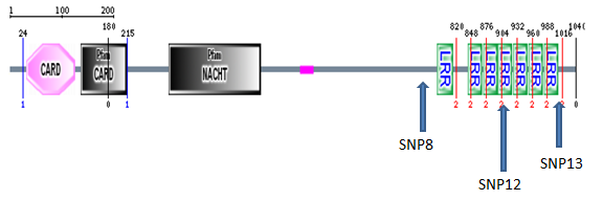
The SMART website was used to search both SMART and PFAM databases for NOD2 protein domains. The resulting domains are as follows:
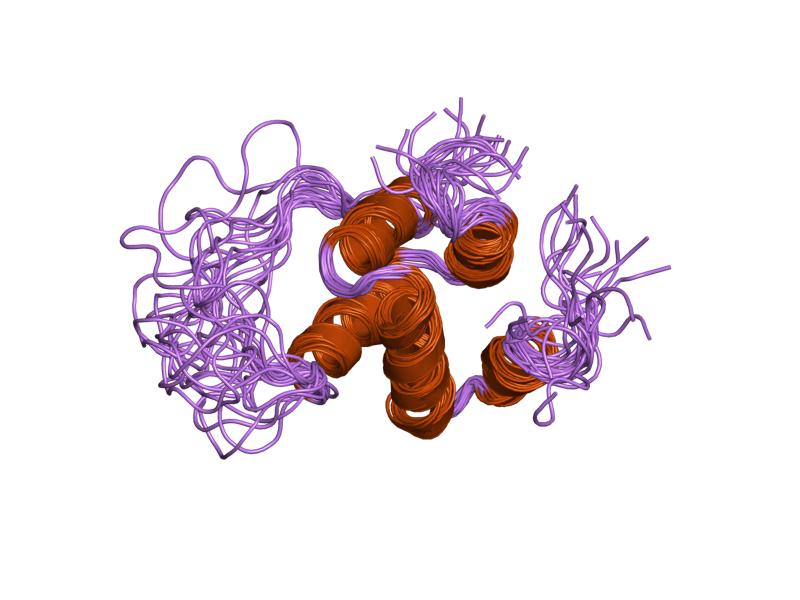
CARD:
Capsase recruitment domains (CARD) are interaction domains made up of six alpha helices. They are often involved in apoptosis as well as inflammation through NF-kappaB interactions [2]. NOD2 contains two such domains at its N-terminal domain. Here they act through RIP2 to ultimately recruit
NF-kappaB to trigger immune response in the gut. In the case of NOD2 these domains are only active when NOD2 is oligomerized with other proteins.
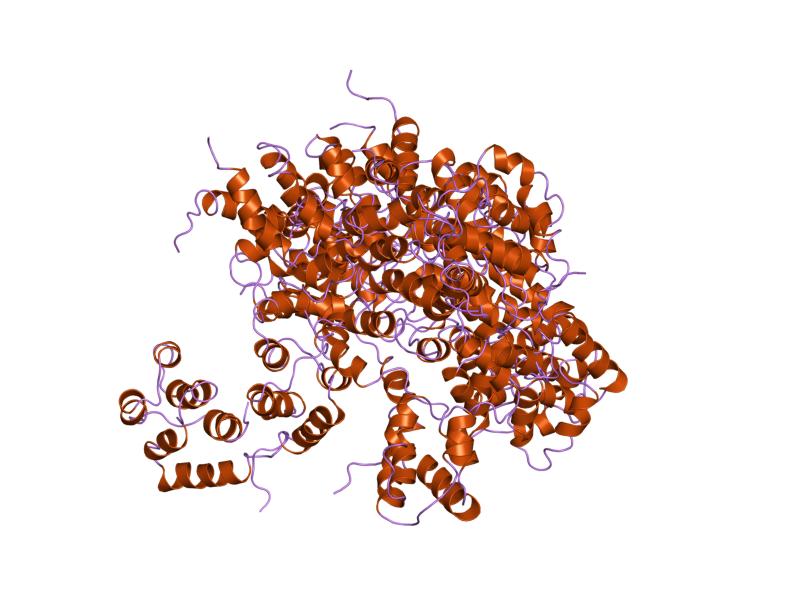
NACHT:
NACHT, a nucleoside-triphosphatase (NTPase) domain is commonly found in apoptosis proteins and those involved in myosin heavy chain transcription. They are also commonly associated with CARD and LRR domains, agreeing well with the presence of three of these domains in NOD2. The Walker B box, a NACHT domain subregion, has shown potential involvement for NOD2-related Crohn's disease. Mutations in this region have been associated with increased autoactivation of the NOD2-associated immune response. Such autoactivation can result in increased inflammation even in the absence of high levels of foreign bacteria [6]. In normal activity, active NOD2 oligomerizes with other active NOD2 through NACHT-NACHT interactions.
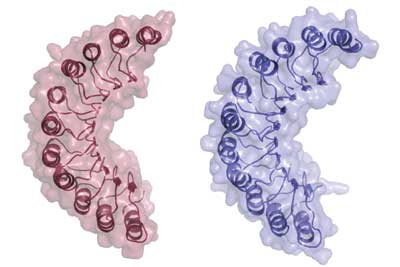
LRR:
Leucine Rich Repeat (LRR) domains are very common protein domains. They consist of alternating beta-sheets and alpha-helices which combine to form a horseshoe structure. They are very important for protein-protein interactions within and between cells. In NOD2 there are 7 LRR domains located at the C-terminal. It has been shown that, in the case of NOD2, these domains interact directly with invading bacteria, leading ultimately to immune response [3]. This region is responsible for detecting bacterial muramyl dipeptide(MDP). After MDP-LRR interaction, NOD2 undergoes a conformational change which allows its NACHT domains to actively bind to other proteins.
Crohn's Disease associated NOD2 mutations
Above I have marked the location of the 3 most common Crohn's associated NOD2 variants with respect to the protein domains. These variants, SNP8, SNP12, and SNP13 are responsible for about 81% of NOD2 associated Crohn's Disease [5].
Analysis
All protein domains present in NOD2 agree well with its function. LRR domains bind directly to MDP and intiate oligomerization via the NACHT domain. Once active and oligomerized, the CARD domains activate downstream response through interactions with proteins such as RIP2 [4]. Of the most interest is the fact that the most common disease associated variants disrupt normal LRR function, and thus disrupt MDP-mediated NOD2 activation. This is consistent with a NOD2 loss-of-function mutation associated with Crohn's Disease.
References
[1] SMART
[2] Bouchier-Hayes L, Martin SJ (2002). "CARD games in apoptosis and immunity". EMBO Rep 3 (7): 616–21
[3] Perez, Laurent-Herve, Matt Butler, Tammy Creasey, JoAnn Dzink-Fox, John Gounarides, Stephanie Petit, Anna Ropenga, Neil Ryder, Kathryn Smith, Philip Smith, and Scott J. Parkinson. "Direct Bacterial Killing In Vitro by Recombinant Nod2 Is Compromised by Crohn's Disease-Associated Mutations." Ed. Niyaz Ahmed. PLoS ONE 5.6 (2010): E10915. Print.
[4] KEGG
[5] Jensen, Stina Rikke, Ole Haagen Nielsen, and Susanne Brix. "Are NOD2 Polymorphisms Linked to a Specific Disease Endophenotype of Crohn's Disease?" Inflammatory Bowel Diseases (2011): Print.
[6] Zurek, Birte. "Functional Analysis of the Nod1 and Nod2 NACHT Domain Reveals Differences in Their Ability to Activate NF-kB." Innate Immunity (2011). Print.
[7] PFAM
[2] Bouchier-Hayes L, Martin SJ (2002). "CARD games in apoptosis and immunity". EMBO Rep 3 (7): 616–21
[3] Perez, Laurent-Herve, Matt Butler, Tammy Creasey, JoAnn Dzink-Fox, John Gounarides, Stephanie Petit, Anna Ropenga, Neil Ryder, Kathryn Smith, Philip Smith, and Scott J. Parkinson. "Direct Bacterial Killing In Vitro by Recombinant Nod2 Is Compromised by Crohn's Disease-Associated Mutations." Ed. Niyaz Ahmed. PLoS ONE 5.6 (2010): E10915. Print.
[4] KEGG
[5] Jensen, Stina Rikke, Ole Haagen Nielsen, and Susanne Brix. "Are NOD2 Polymorphisms Linked to a Specific Disease Endophenotype of Crohn's Disease?" Inflammatory Bowel Diseases (2011): Print.
[6] Zurek, Birte. "Functional Analysis of the Nod1 and Nod2 NACHT Domain Reveals Differences in Their Ability to Activate NF-kB." Innate Immunity (2011). Print.
[7] PFAM